The following manuscript was written in the late 1970s but never published.
IS IT POSSIBLE TO CONTROL POLLUTION?
R. F. Mueller, December, 2005
SUMMARY
The driving force of active pollutants of any kind results from their excess free energy relative to their innocuous degradation products1. The great variety of industrial processes and pollutants results in the statistical equivalence with respect to degree of deleteriousness of various technological energy sources in their utilization and dissipation modes, and this deleteriousness depends on the total culture. Thus, "clean" energy sources such as solar energy ultimately give rise to the same distribution of pollutants as "dirty" sources. Similarly, the concepts of "soft" and "hard" energy paths have no meaning in a thermodynamic sense.
Because all thermodynamically possible technological processes must be utterly spontaneous in sum, there can be no overall control of energy or the equivalent pollution. In the installation of pollution control equipment this takes the form of an additional increment of energy and hence pollution, which is added to that already resulting from the primary industrial process. Thus, any control exercised over local pollution generally results in pollution elsewhere.
INTRODUCTION
This piece might just as well have been entitled "Is it Possible to Control Energy?" Certainly the connection between energy and pollution, seemingly misunderstood or at least obscured, in one way or another, lies behind the plethora of problems that are actively eroding the benefits of modern technology and are calling this technology itself into question. This state of affairs has encouraged the growth of unscientific myths, which not only have captured the public consciousness, but continue to be spread by the highest scientific authorities, and most particularly by those in the ranks of technological optimists2. Anticipate then that I regard the "control" of energy and pollution by technologic means as one of these myths, which Cambel3 conveniently phrased as follows:
"The solution to the conflict between energy and the environment must not be in curtailing energy supply, but in reducing the irreversible and dissipative effects when we convert and consume energy."
Or, in what is in part a corollary to this viewpoint, no less than a presidential science advisor4 stated:
"We know that some time in the next century we will achieve a virtually unlimited, renewable and clean energy supply."
Even energy-environment specialists have joined in. However Odum5 expressed some ambivalence when he wrote:
"With new, rich energy flows man can control and manage enormous quantities of the world's energy. Increasingly he is affecting the whole biosphere." (italics added)
It is not my aim here to argue that control of energy and pollution on even a quite large scale is precluded, although this may well be the case. It is also well to clarify that what is meant here is control through technology, which is quite a different thing from the obvious methods of administrative control. We have, on the basis of limited evidence, no reason to doubt the instances in which control technologies are said to have resulted in notable improvements in substantial air and water masses. For example, the Presidents Council on Environmental Quality has documented recent decreases in concentration of carbon monoxide and other toxic air pollutants in a number of American cities as a result of emission controls. Also, concentrations of such water pollution indicators as coliform bacteria and oxygen-demanding materials have shown decreases in some streams after sewage treatment was upgraded. Other examples could be cited6 . However, these technological success stories only highlight the question of the efficacy of technology in solving problems which it gave rise to in the first place and which require inputs of additional energy. We should always bear in mind the simple logic that it takes energy to control energy. In any case, the appalling scale of certain emerging pollution problems such as acid rain and toxic organic chemicals should make us suspicious that local controls conceal larger regional effects.
It is clear that if our local pollution control efforts, be they yet so successful, do not result in an overall decrease of pollution in qualitative and quantitative terms, or result in a worsening of global conditions, they are little more than hoaxes perpetrated on ourselves and posterity. Secondly, it is obvious that to the present these large scale effects have not really been studied by those who advocate technological solutions. In a sense this failure is understandable given the complexities not only of technology itself but also because of the need to consider economic and social factors. For example, in matters of economics of life styles, certain effects such as increased employment increments resulting from the initiation of pollution control strategies are usually touted as desirable side effects, when in reality they contribute to undermining the original objective (see examples later!). However, technocrats generally behave as if such problems do not exist or have been solved.
The complexities inherent in the assessment of energy intensive technology may prevent the assembly of adequate data or even our knowing precisely what data are required. Another approach is open to us, however, since the same physical laws apply here as have been so successfully applied in laboratory experiments and in industrial processes, and some of these laws are broad enough in scope to enable us to draw certain conclusions and to eliminate certain alternatives based on the general nature of energy transformations, despite the missing details. Foremost among these systems is the classical thermodynamics.
This science found its first applications in the explanation of precisely those industrial processes that still are central to our environmental dilemma and which appeared with the machines of the early industrial revolution. We recall that Benjamin Thompson (Count Rumford) first surmised the conversion of mechanical energy to heat in the process of boring a cannon, and at the same time dealt a fatal blow to the caloric theory7 . This insight was followed by the precise experiments of Joule and others, who also began the erection of the theoretical framework that culminated in the work of Gibbs. While this approach led to ever more sophisticated applications in industrial processes and in pure science, it was, strangely, never extended to technology as a whole, nor has there been any widespread interest in obvious applications to environmental problems which have their source in this technology. Attention should however be paid to the efforts of Odum5 , Miller8 and, in the related field of economics, Georgescu-Roegen2. With the exception of such rather isolated examples, an impression is gained that, while thermodynamics applies to individual machines, it does not apply to the sum of the effects of all machines or the general energy flux through society. With the recent threat of energy shortages there has to be sure developed a considerable interest in various forms of energy "bookkeeping", in which agricultural and industrial processes and the means of energy production have been studied exhaustively for their efficiencies. A good example is the work of Kakela9 , which purports to show support for the technological optimist's view that a resource can be indefinitely extended through technological progress. However, these studies seldom are concerned with tracing energy use beyond the industrial process itself or with entropic effects. While this approach has led to some useful results and toward more quantification, it is not true thermodynamics, the application of which may actually enable us to dispense with much quantification of this type.
The approach here also differs in some important ways from those of Odum, Miller and Georgescu-Roegen, since these authors seem to have at least a residual faith in the solutions of high technology. Although this impression is most apparent in the work of Odum, Miller looks with hope to a sort of cybernetic age in which information handling plays the paramount role. Elsewhere, however, he states that"...there is no technologic solution to pollution." Generally Georgescu-Roegen is very forceful in stressing the overriding power of entropic limits to technology. Yet all these authors in one way or another hold out hope that humankind will discover the elixir of a source of clean, abundant technological energy-generally regarded as solar in origin. Here, however, the reader will find this hope denied by more than skepticism, beginning with a concise definition of pollution as energy, as well as the argument for the ultimate statistical equivalence in terms of deleteriousness of all forms of technologic energy, whether they are "clean" or :dirty" in origin. In this way a clean break is made with previous viewpoints on this subject.
Figure 1: Energy use and waste in a generalized machine.
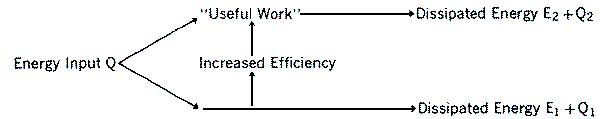
Figure 2: Energy dissipation as a result of technological processes.
RELATION BETWEEN ENERGY AND POLLUTION
The unsavory relation between technological energy and pollution has of course been widely recognized, but almost invariably it has been assumed that technologists are crafty enough to cure the maladies technology itself has engendered. This attitude is quite apparent in the quotations cited earlier in which environmental problems brought on by glutinous consumption are regarded as avoidable or at least reducible to manageable proportions. Can this be true? Can the fox really regurgitate live hens?
To examine this question it is helpful to begin by generalizing the concept of a machine, which we may define as any technologic device or process intended to achieve some desired result through the expenditure of energy. This generalized machine, depicted in Figure 1, consists of a box in which a certain quantity of delivered fuel energy Ef is converted into "work" W, with an intended result R, and from which a quantity of wasted energy E1 + Q1 is emitted, where E1 is energy stored in the environment, perhaps temporarily, and Q1 is the dissipated heat.
The energy symbol E represents the familiar thermodynamic state function, which usually consists of internal chemical energy of constitution, but may also include such forms as gravitational energy. Q and W are never state functions but depend on the path of application.
A point of some significance here is that not only is there an unavoidable waste energy E1 + Q1 , depending on machine efficiency, but also the "useful work" W must inevitably be dissipated in its entirety as pollution E2 + Q2 , in what might be called the "Rumford Effect." Thus the environmental side effects of energy utilization extend far beyond energy production and industrial use to consumer use and "wear-out", in which the energy of constitution, in indeterminate but usually deleterious modes, interacts in most intimate ways with organisms, and in particular with humankind. This happens, for example, when asbestos contaminates such consumer products as baby powder, or when tissue-reactive hair dyes are used. Although such cases are recognized as pollution, they are not usually considered as resulting from the dissipation of technological energy.
It is a refinement to point out that sometimes a quantity of energy is stored in R, as when work is expended in lifting a mass. However, frequently this is not the case, and R does not represent a higher energy state than the preexisting one. In fact, R may represent a lower resultant energy state for a product than a previous one. In the case of no energy gain, the energy has been expended on the environment, as in much transportation, in which it is dissipated as friction or as other forms, and does not reside in the mass.
The operation of a machine is most easily perceived in the common cyclic engine in which Ef is first transformed into heat Q. In this case the work is derived from Q, and Q1 is rejected into the environment as waste. The thermal efficiency of such a machine is
(W/Q) = (Q-Q1 )/Q,
which is strictly limited by the difference in temperature between the heat source and sink and cannot exceed the efficiency of the ideal Carnot engine. An important point here is that in addition to the initial dissipation of the heat Q1 the dissipation of W as E2 + Q2 is indispensable to the operation of the machine, as when friction between the wheels and ground is required to move a vehicle.
It is interesting to consider a quite different example, the expenditure of a chemical pesticide to kill some target organism. In this case also there is an initial input of chemical energy Ef , which enters the habitat of the target. Some of this energy impinges directly on the organism and is analogous to mechanical work. However, the greatest part of the energy misses the target and interacts with non-target organisms or other substances of the environment. In this process some energy storage occurs and some heat is released through the chemical reactions which occur. Thus again E1 + Q1 is dissipated. However, even that part of the pesticide energy which reaches the target organism continues to interact with the environment and non-target organisms as partially degraded (in part metabolized) but still reactive forms of the original chemical. This is E2 + Q2 .
At this point it is helpful to introduce additional thermodynamic state functions, the entropy S, and the Gibbs Free Energy G. The entropy function is important because it incorporates the effect of the Second Law of thermodynamics. G simply represents a convenient artifice because it includes frequently occurring combinations of more familiar variables of the system under consideration. Thus we have the fundamental relation
Gsy = Esy - TSsy + PVsy (1)
in which P is the pressure, V the volume, T the temperature in degrees Kelvin, and E is the energy as previously defined. The subscript sy indicates the system under consideration as distinguished from the surroundings.
The justification for introducing Gsy is that Esy, the energy of the system, does not necessarily change (increase or decrease) in any indicative manner in a given process, while Gsy invariably decreases if the process is physically realizable ( spontaneous). Thus, the change in free energy ΔGsy < 0, provides an index of great utility in environmental problems, as it does in conventional technologic and scientific applications. On the other hand, ΔSsy, as well as ΔEsy may be zero, negative or positive in a given spontaneous process, depending on circumstances. However, ΔSsy is related to the total or net entropy change ΔST and the entropy change of the surroundings ΔSsu as follows:
ΔSsy + ΔSsu = ΔST . (2)
Since, by the Second Law of thermodynamics, ΔST must always be positive for any spontaneous process, it, like ΔGsy , provides a useful index of such a process. The choice between ΔGsy and ΔST is a matter of convenience in practical calculations, since values of the former are frequently tabulated, or a matter of taste in theoretical discussions, with the latter preferred by some authors such as Georgescu-Roegen2 , for example.
As an illustration of the critical role of energy in environmental degradation we consider the notorious pollutant carbon monoxide, which may also react with atmospheric oxygen as follows:
| Carbon monoxide | oxygen | carbon dioxide | |
| CO | +1/2 O2 | ===> | CO2 |
Under the standard temperature and pressure (STP) conditions (T= 298o K, PO2 = PCO = PCO2 = 1 atmosphere), which serve as a basis of reference, ΔGsy for the above reaction is -61,650 calories10. Whether CO reacts in this innocuous manner in the open air or in a deleterious manner within an organism, of course depends on where the reactive molecule finds itself. The point here is that in a thermodynamic sense the reaction occurs because the reactants (CO and O2 in this case) have excess free energy relative to the product ( CO2 in this case).
The foregoing example illustrates the universal characteristic of chemical pollutants by which they possess free energy in excess of that possessed by their degradation products and thus defines the fundamental relation between energy and pollution. Of course, degradation is usually step-wise, and in this case although CO2 may be regarded as less noxious than CO by some, it should be remembered that the former also has a potential deleteriousness in that it enhances Earth's greenhouse effect and may, in time, bring about disastrous global warming. However, further degradation is possible, as when CO2 reacts with crustal rocks to form such stable carbonates as MgCO3 and CaCO311 , which for all practical purposes eliminate this constituent as any form of pollutant. Unfortunately, the latter reactions are too slow to be of much help.
Many other examples could be cited. Thus, the common stack emissions of fossil fuel-fired power plants contain sulfur dioxide, which reacts with atmospheric and stack oxygen and water to yield highly corrosive sulfuric acid, which not only injures living tissue, but attacks metals and even silicates of building stone 12. Recently far-flung deterioration of fishing waters and vegetation from this source has been recognized in acid rain13.
Excess free energy is not confined to chemical reactants, but is a universal property of all pollutants.
It manifests itself in such diverse cases as silt pollution of bodies of water, in destructive erosion of soils, and in toxic effects of certain microscopic particles. For example, "pure" water falling on soils of natural areas does not behave as a pollutant because it has not been excessively energized. Yet, through human activity it may become a pollutant. If vegetated lands are deprived of their cover and mechanically disturbed, as in timber cutting or overgrazing, their soils are eroded and transported by more rapidly flowing and hence energetic water, whose excess energy is then transferred to the soil particles. Even clear water energized by flowing over technologically smoothed surfaces such as pavements may be considered a pollutant since the excess kinetic energy it thus acquires will later be spent in eroding stream beds and banks. As distinguished from technological energy which has its source in fuels, this form of pollutant energy has its source in natural energy flows which have become misdirected through far smaller "activating" technological energy inputs14. Such deleterious hydrologic effects are at the heart of worldwide problems of desertification, flooding and famine.
The most familiar example of the toxic particle effect is the hazard of the mineral asbestos. The toxicity of asbestos probably results from excess free energy which it acquires during processing when the mechanical separation and fracture of fibers results in numerous poorly compensated charges on the fiber surfaces and edges. These charges would tend to react readily and deleteriously with contacting tissues much as do ions produced by radiation.
If, as we have inferred, all forms of pollution are characterized by excess free energy, does it then follow that all energy ends up as pollution, corresponding to energy = pollution? The answer to this question is to be found in the evolutionary history throughout which organisms have become adapted to specific types and spectral ranges of energy. As an illustration, visible light energy is obviously the opposite of pollution to green plants, while excess ultraviolet or gamma rays would constitute obvious pollution. Unfortunately, nearly all technological energy corresponds to pollution due to this maladaptation quality factor. At this point it is also well to stress that this factor is at the heart of the degree of deleteriousness which different forms of energy of identical quantitative measure possess. Elsewhere14 the writer has indicated the need to make general allowance for this differential deleteriousness in a factor analogous to the "quality factor" already in use in dealing with radioactive substances.
An entirely different way in which energy of pollutants manifests itself is through the process by which these substances become dispersed in the environment. If, for example, we consider a hypothetical toxic solid A that becomes an active pollutant by dissolving in water, we may write the solution reaction as
Solid Solution
A ===> A
To simplify the illustration we assume that A forms an "ideal" mixture with water11. The change in Gibbs Free Energy on solution may then be written as
ΔG = ΔGo + RTln XA (3)
where R is the universal gas constant, ΔGo is the Gibbs Free Energy change under standard reference conditions when XA= 1, and lnXA is the natural logarithm of XA, the mole fraction concentration of A in the water solution. It is obvious that as XA ==> 0, corresponding to high dilution or "pure" water, ΔG assumes highly negative values, or ΔG << 0 for any constant value of ΔGo, so that there is developed an enormous driving potential to bring the toxic substance into solution. We note that for any constant value of ΔGo this potential results solely from the entropy of mixing term -RlnXA , which is an important contribution to the negative free energy change.. This type of entropy change is always positive and provides a measure of the tendency of molecules A to "get lost" among the water molecules. It is this effect which is behind much chemical pollution and which makes it so difficult to prevent and clean up. It is also a wonderful illustration of the physical constraints on resource inputs in the economic process, as has been stressed by Georgescu-Roegen2 . Because of this effect resources which are originally dilute, or which have been irreversibly dispersed through use, become prohibitively expensive to recover, and no industrial process can be designed to circumvent it. An attempt in this direction was made by Kakela9 , who used as an illustration the energy and labor savings which result from a shift from high grade to pelletized low grade iron ores. However, in this case the savings are attributable to the fortuitous textural and magnetic properties of the latter. His analysis also ignores the component of energy dissipation which in mining these ores results in serious pollution, especially by amphibolitic asbestos15.
THERMODYNAMICS OF TECHNOLOGICAL DEVELOPMENT
We note first of all that nature does not distinguish between the primary activities of processing, manufacturing, etc., which supposedly benefit humanity and which give rise to environmental problems, and those secondary activities, such as recycling and pollution control, which are intended to solve these problems. Both are subject to the same limiting thermodynamic laws. In this sense there is no difference between a polluting factory and the pollution control device intended to abate pollution, no difference between the so-called benign or "soft" technology that is hailed as a panacea by some environmentalists16 and its "hard" counterpart. The same argument applies to the concepts of "clean" and "dirty" technological energy as usually distinguished. Furthermore, I will try to show that the concept of technological efficiency, which is touted as one of the cures for industrial pollution, loses its meaning if the environment is factored in.
We have seen that the manifold pollutants have the one common characteristic of possessing excess free energy relative to their degradation products. This energy is either 1) inherent in the raw materials (such as fuels), 2) is conferred by processing or manufacturing or 3) results inadvertently from a variety of human activities.
The efficiency of a machine may be illustrated as in Figure 2, which shows how this may be increased by diverting a greater proportion of input energy to "useful work." However, we have already noted how uncontrolled dissipated energy E2 + Q2 results from degradation of this work. This disposes of the idea that pollution is solely the result of "waste." We note first of all that there is no intrinsic reason why E2 , Q2 etc. should be less deleterious in nature than E1, Q1 etc. Also, if we imagine that an increase in efficiency results in a new distribution E'2, Q'2 etc., we have no reason to hope that in general deleteriousness will decrease per unit of energy dissipated. Finally, we note that on average different energy sources will, in the same culture, result in the same general distribution of pollutant energy forms, and hence degree of deleteriousness. Thus hydro or solar electric energy will be used to produce identical toxic chemicals as does that of nuclear electricity. The great variety of energy conversions in modern technology assures a certain statistical equivalence of deleteriousness in each unit of energy utilized, regardless of source, and the average degree of deleteriousness is a function of the total culture at a given moment. Consequently, the environment on average sees only Ef or Q, the total energy input. In this context the only benefit to the environment of increasing the efficiency W/Q is that such an increase will require a smaller Q or Ef to accomplish a given task. In other words, the environmental impact of a machine depends primarily not on W/Q, but on Q. From this result it may be concluded that no matter how efficient industry becomes, continuous expansion can only bring increasing pollution and environmental deterioration.
The indeterminateness of the E's and Q's with respect to deleteriousness results from the same Second Law constraints that impose quantitative limits on W/Q and is an expression of the characteristic that all energy transformations are utterly spontaneous. Consequently there can be no overall control of energy release or utilization. Energetically all that is accomplished in the industrial process is the "arrangement" of energy events such as useful chemical reactions in a manner analogous to the arrangement of a party or celebration. However, the energy party always proceeds according to its own rules (which an invention discovers) and usually feeds on larger and more remote energy systems whose very existence is sometimes unsuspected. For example, we should question who monitors all the pollution that results from a pollution control program!
Although the Second Law effects form the basis of machine inefficiencies and environmental impacts, they are also vital to its very operation. Thus the increase in entropy and the energy loss associated with friction are not only necessary to such processes as locomotion, but act to dissipate the energy after it is utilized. For example, friction between wheels and ground is required to move a vehicle, but the resultant heat must be dissipated to prevent undue rise in temperature. Similarly, in the case of a chemical pesticide, the increase in entropy associated with the spontaneous dissemination and dissolution of the pesticide, as described by equation (3), is a requirement for reaching the target organism, although it is also an index of inefficient use and results in undesirable impacts.
Now consider the effect of the application of a pollution control device on an industry from the standpoint of the First Law requirements on energy flows. First consider the situation without pollution controls, in which a quantity of energy E1 is applied to raw materials M1 , yielding a useful product M2 and pollution P1. The Ps and Ms incorporate the same internal energies of constitution as the Es and are expressed in the same units. However, implicit in them are other energy forms as well. Thus the Ps may be regarded as equivalent to E1 + Q1, while the useful product dissipates to E2 + Q2 as discussed earlier. The pollutants individually and collectively are subject to the constraints of equation (1), as expressions of the Second Law, which also limits control over the degree of deleteriousness.
If pollution control energy Ec is applied to pollution P1, the latter is thereby reduced to P'1. Note, however, that by the First Law, the "controlled" component P1 - P'1 has not disappeared, but has merely been transformed and/or transported elsewhere. Furthermore, the added increment Ec = P'1 + P2 - P1, where the total dissipated energy of pollution control, represented by P2, must now be disposed of. For example, the sludges generated by certain pollution control devices may be transported to disposal areas where they bring blight to the landscape and poison water supplies17, or SO2 vented in tall smokestacks may appear hundreds of miles downwind as acid rain. Proposals for the reclamation of useful chemicals from such sludges and emissions would of course increase the increment Ec. The important point, however, is that P2 + P'1 > P1, which is the inevitable result from the laws of thermodynamics. Also, as we have seen, the statistics of the diverse Ps results in a rough equivalence in their degrees of deleteriousness. Generally, however, one pollution form is favored over another for cultural or other reasons, resulting in tradeoffs which may not always be wise. For example, the immediate benefit of achieving clean sewage plant effluent is usually deemed more important than assurance of the safe disposal of resultant sludges, which may eventually poison ground water. A more suitable effort would entail the low energy solution of prohibiting toxic materials such as heavy metals, pesticides and household chemicals from entering the sewage system in the first place.
CONCLUSIONS
The monstrous character of nuclear energy is brought home to us by a quotation from Weinberg18 to the effect that nuclear technologists have made a Faustian bargain of sorts with society, the chief problem being the long term "control" necessary in safeguarding nuclear wastes. However, the analogy does not go far enough in that the "bargain" is seen here as an illusion of control which pervades all of energy intensive technology.
Although it is difficult to do a sufficiently thorough bookkeeping job to uncover all the energy input and dissipation modes of even a simple technology, some highly suggestive indicators exist. From the literature of "pollution control", for example, there emerges a sequence of cases of varying degrees of obviousness of the breakdown or failure of control. Rather than discuss the most obvious cases, which, for example, include that of a technology not achieving previously set standards, we pass immediately to that of greatest interest, in which completely effective pollution control is apparently achieved, but, as has been true so far, external interactions of the control technology have not been taken into account. The indirect form of pollution envisioned stems here not only from the complexities of the technology itself, but to a great degree from its societal underpinnings, from the many connections and lifestyle ramifications still mostly unappreciated. The statistical equivalence of pollution and technological energy argued for earlier provides a convenient bridge to studies such as those of Hannon19 and Herendeen20 , which relate the degree of energy intensiveness to economic factors and life styles. According to these studies there is an almost linear relation between total consumer ( family) energy use and income, so that regardless social stratum, total energy use rises directly with income. We need only substitute "pollution" for "energy" in this relation. Not surprisingly it was found by them that the most labor intensive activity ranked lowest on the economic scale.
The bearing of all this on the effects of pollution control technology is as follows: It is apparent first of all that any technology, no matter how sophisticated, has numerous links to basic extractive industries, support industries, etc. that are usually highly pollution prone in their own rights. Thus a pollution control technology may itself ultimately depend on rather primitive mining industries as well as fossil or nuclear fuels that emit a host of toxic pollutants, albeit in remote locations. More interesting, however, is the supporting societal infrastructure. For example, it is a common boast of the environmental optimist that one of the benefits of pollution control programs is that they "create jobs." According to an article by Green21 , the US Environmental Protection Agency estimates that the number of jobs directly resulting from pollution control expenditures is as high as 600,000. Similar figures are quoted for West Germany. Now we know that many of these jobs are of a highly skilled nature and pay well, so that they fall high on the Hannon and Herendeen curve. Thus pollution control must give rise to the same two classes-production and consumption- of pollution sources, and of the same order of intensity, as does ordinary consumer activity in an affluent society. The pollutants thus generated are of course an integral part of P2.
It must also be concluded that if the evolution of society as a whole is considered, there appears to have been no real progress in the efficiency of energy use or in stemming waste and pollution from this source. There can of course be no doubt that, taken by themselves, the efficiencies of modern factories with respect to energy and material use greatly exceed their earlier counterparts22 . Yet the progressive use by industry of more dilute (lower grade) material resources imposes an inefficiency of its own on total resource use that counters increases in the machine efficiency which makes this use possible. For example, the mining of low grade ores made possible by new machines and techniques creates more waste and pollution per ton of metal recovered than ever before2 . Consequently, the total effect of mining activities and those industries which depend on them is constantly decreasing efficiency. Also, the advent of ever more technologically elaborate and sometimes more energy efficient machines in the consumer society has given rise to corresponding increases in trivialization, which, in a pragmatic sense, is inefficiency. The smoky but practical passenger trains of the 1930s were replaced by automobiles traveling over highly impractical routes (suburban commuting etc.) or for entirely trivial reasons. Similarly, the recycled milk bottles and other containers of this early period yielded to the "throw away" system of today.
On the other side of the coin we have seen that ever increasing entropy associated with the ever expanding technological energy flux is synonymous with disorder and loss of control. Is it possible then that there is a simpler relation between energy use and social problems than anyone suspected? Is there, despite the elaboration of technology, an inevitable decrease in social order which results from this massive societal energy flux that can no longer simply be rejected into the "surroundings." Certainly the disordering effects of superimposing high energy economies and social mores on traditional societies such as those of the Middle East are apparent. Even the fact that corresponding social disorder does not appear in even higher energy established industrial societies, does not contradict this thesis, since the latter may have achieved an at least metastable adaptation of sorts during the comparatively slow evolution to the high energy flux of their own industrializations.
We may now summarize our answer to the questions raised in the introduction. First, is it possible to control pollution? If we mean by this many of the commonly accepted controls through technology, the answer must be an emphatic no. According to thermodynamic reasoning, we can order or "arrange" a part of the local environment only at the expense of disordering the whole. Thus, local pollution control, which depends on energy inputs, can only result in greater pollution of the total (global) system. This result does not of course preclude pollution control through such means as technological innovations or administrative actions which decrease energy inputs. Thus we can effectively ban a pollutant such as a pesticide, shut down an industry, or substitute a more efficient technology. Of course such actions may have their price in loss of convenience or jobs or in increases in the price of goods.
Secondly, is there really an entity like "clean energy'? According to the foregoing analysis we must conclude that clean technological energy is a contradiction in terms. Once an energetic process is initiated, the process itself determines the energy path of dissipation and the resultant pollutants, unless additional energy, with its own burden of pollutants, is brought to bear through control technology. In this way energy produced by so-called "clean energy sources" is immediately converted to the societal mix of dirty energy forms. The same reasoning applies to the concepts of "hard" vs. "soft" technologies.
We see then, that as far as the foregoing analysis is concerned, there is nothing whatever to encourage the technologic optimist who favors the type of economic growth that depends on ever increasing energy inputs. Indeed, as Georgescu-Roegen2 has pointed out, there is little more solace in the steady state economy, since this at very least requires a steady input of energy, which may be very large for large populations. There is also very little to buoy up the spirits of environmental optimists, who tend not only to favor a steady state, but also retain a naive faith in the possibility of pollution "control" by means of technological devices. Such are the enigmas and dilemmas we now confront. To them sophisticated analysis may indeed bring insights, but only self-denial and austerity can bring solutions.
NOTES AND REFERENCES
1. Mueller, R. F. Thermodynamics of Environmental Degradation. National Aeronautics and Space Administration Document X-644-71-121, 1971.
2. Georgescu-Roegen has discussed analogous myths which infect economic thought: Georgescu-Roegen, Nicholas, Southern Economic Journal 41, 347, 1957.
3. Cambel, A. B., Physics Today 23, 38, 1970.
4. Press, Frank, Science 200, 731, 1978.
5. Odum, H. T., Environment, Power and Society. John Wiley & Sons, Inc., New York, 1971.
6. Council on Environmental Quality, Executive Office of the President "Environmental Quality, The Ninth Annual Report of the Council on Environmental Quality." 1978
7. Faires, V. M., Thermodynamics. The McMillan Co., New York, 1962.
8. Miller,G. Tyler, Jr., Energetics, Kinetics and Life. Wadsworth, Belmont, California, 1971.
9. Kakela, P. J., Science 202, 1151, 1975.
10. Rossini, F. D., Pitzer, K. S., Taylor, W. J., Ebert, J. P., Kilpatrick, J. E., Becket, C. W., Williams, M. G. and Werner, H. G. Selected Values of Properties of hydrocarbons. National Bureau of Standards Circular 461, 1947.
11. Mueller, R. F. and Saxena, S. K. Chemical Petrology with Applications to the Terrestrial Planets and Meteorites. Springer Verlag, New York, 1977.
12. Council on Environmental Quality, Executive Office of the President "Environmental Quality, The Eight Annual Report of the Council on Environmental Quality" 1977.
13. Galloway, J. N., Likens, G. E. and Edgerton, E. S. Science 194, 722, 1976.
14. Mueller, R. F., Science 192, 11, 1976.
15. Council on Environmental Quality, Executive Office of the President "Environmental Quality, The Seventh Annual Report of the Council on Environmental Quality." 1976.
16. Lovins, A. B., Soft Energy Paths: Toward a Durable Peace. Friends of the Earth-Ballinger, Cambridge, Mass., 1977.
17. Hammond, A. L. Science 194, 172, 1976.
18. Weinberg, A., Science 177, 27, 1972.
19. Hannon, B., Science 189, 95, 1975.
20. Herendeen, R. A., Am. Soc. Mech. Eng. Tech. Digest 73-WA/ENER-8.
21. Green, Mark, Environmental Action 10, 22, 1979.
22. Berg, C. A., Science 184, 264,1974.