THERMODYNAMICS OF ENVIRONMENTAL DEGRADATION*
Robert F. Mueller
Planetology Branch
Goddard Space Flight center
Greenbelt, Maryland 20771
March 1971
*Presented at the Annual Meeting of the American Geophysical Union, Washington, D. C., 1971.
GODDARD SPACE FLIGHT CENTER
Greenbelt, Maryland
| CONTENTS | ||
| INTRODUCTION | .................................... | 1 |
| POLLUTION DEFINED | .................................... | 1 |
| THERMODYNAMICS OF POLLUTION | .................................... | 3 |
| ENERGY CONCENTRATION IN MODIFIED REGIONS | .................................... | 8 |
| RECYCLING, POLLUTION CONTROL AND LIFE STYLE | .................................... | 8 |
| ECONOMICS AND THERMODYNAMICS | .................................... | 10 |
| SUMMARY AND CONCLUSIONS | .................................... | 11 |
| REFERENCES | .................................... | 13 |
| ILLUSTRATION | ||
| Modifications of Natural Region by Technology | .................................... | 6 |
THERMODYNAMICS OF ENVIRONMENTAL DEGRADATION
INTRODUCTION
Environmental degradation today has assumed the magnitude of planetary processes such as geologic erosion or the atmospheric circulation. Understanding of the many facets of this degradation in scientific terms will probably exhaust most known disciplines.
It is the thesis of this paper that the laws of thermodynamics, which are concerned with energy transformations, place severe constraints on the affairs of man and his relationship to his environment. Indeed, thermodynamic principles seem particularly well suited to treating the interaction of technology and natural systems and the major product of this interaction, pollution.* In this light the recently enunciated laws of ecology (Commoner, 1969) are seen as the biological and sociological expressions of the thermodynamic laws. We shall see that both ecology and thermodynamics are also closely paralleled by new viewpoints of economics (Olson and Cumberland, 1970), which are emerging as a result of environmental concerns.
This paper is concerned with the general features of environmental thermodynamics and attempts to set forth the postulates which are regarded as necessary for a more detailed and quantitative development.
POLLUTION DEFINED
In a sense all pollution may be regarded as symptomatic of the general degradation of Earth's natural^ realms- air, water and land. As these are converted to man's technological use the environment is degraded in numerous ways. It should be made clear at the outset however that natural processes such as forest fires also pollute. There is little difference in kind between these and technological sources of pollution. What is different about technology is the magnitude of its impact and the characteristic that the organic world contains limited genetic resilience to respond to technological innovations of this magnitude.
*The operation
of planetary processes such as the formation of lithospheres,
hydrospheres and atmospheres are best understood through
thermodynamic analysis. Conversely, many of the broader aspects of
environmental problems may be regarded as falling within the purview
of planetology.
^While the
shortcomings of this term are recognized, it was thought best to
adhere to well established terminology. As used here "natural"
refers simply to phenomena which predate man's technology.
The comparison and summation of the total effects of pollution are hampered by the diversity and virtual lack of common characteristics of the many pollutants. It is difficult, for example, to compare the effects of sewage pollution and sediment pollution, or of either of these with the effects of pesticides. However, there is one characteristic which all pollutants appear to have in common and which serves to define them in thermodynamic terms: their high energy content relative to the raw materials from which they were derived and relative to the degradation products which they ultimately yield. Their high energy content relative to the latter is particularly apparent since this energy must be dissipated in the spontaneous process of degradation. It may turn out that the energy content of pollutants is a reliable index of their environmental impact.
Through man's not always purposeful intervention natural materials acquire energy in excess of what they would possess had this intervention not occurred. In this way practically any material may become a pollutant. We may illustrate by considering an example from hydrology. "Pure" water and soil or subsoil in natural areas do not behave as pollutants. Yet through man's activity both may become pollutants. If naturally forested land is deprived of its vegetation and mechanically disturbed, not only is the topsoil lost, but also the subsoil is eroded and transported down stream. On such land the energy of falling water is not dissipated through absorption by the leaf mat and topsoil. Rather the water moves downhill in sheet wash or in temporary streams. During flow this water also acquires the potential energy of the disturbed soil particles. The resulting turbid waters deposit sediments that clog stream channels and otherwise diminish environmental quality. Consequently, silt is widely recognized as a major pollutant (The Cost of Clean Water, 1969). However, even energized pure water with no sediment load may be considered a pollutant since its excess energy will later lead to erosion of stream beds and banks.
In another example, a wide variety of chemical pollutants are characterized by the possession of free energies of formation in excess of those of the raw materials from which they were derived. This energy was of course purposefully conferred through the manufacturing process and is again released as the material goes to a lower free energy state in spontaneous degradation. A good example is provided by the native metals which release heat (decrease in enthalpy) as they combine with oxygen (Kubaschewski and Evans, 1958). A quantitative illustration of the effect is also afforded by the SO2 production of fossil fuel power plants. The subsequent step-wise degradation of this component gives rise to numerous detrimental effects in the environment. A 350 megawatt plant vents approximately 75 tons of SO2 per day (Urone and Schroeder, 1969). In sunlight SO2 yields a variety of photochemically induced products. However, much of it is ultimately converted to H2SO4 (sulfuric acid) according to the overall reaction
| gas | gas | solution | solution | |
| SO2 | + 1/2 O2+ | H2O | ==> | H2SO4 |
The standard enthalpy change for this reaction is -78,053 calories. If all the SO2 from the 350 megawatt plant were converted to H2 SO4, the enthalpy change would be equivalent to 4.4 megawatts. Of course, the sulfuric acid produced would continue to react with its surroundings. A typical reaction of this type is
| Metal | gas | solution | crystalline | solution | |
| 2Fe+ | 3/2O2+ | 3 H2SO4 | ==> | Fe2(SO4)3 | + 3 H2O |
However H2 SO4 attacks a variety of other substances, including silicates (Raymahashay, 1968), and so results in considerable economic loss. Some of these degradation processes may appear as toxic effects in organisms. In the case of pesticides these energy dissipative processes are purposeful, but in many other environmental contaminants they are not.
However, we must not confuse the thermodynamic stability of a chemical compound with any resistance it may show to degradation. DDT is persistent in time, not because it is thermodynamically stable, but because it is kinetically stable. It simply takes a long time to react with its surroundings.
THERMODYNAMICS OF POLLUTION
- Gradual build-up of pollutants
in the modified region
- Geologic, soil and climatic
evolution
- Biologic evolution
When a heat engine yields "useful work" through the consumption of a given quantity of fuel energy, a certain quantity of heat is always rejected to the external environment. Thus if Q1 is the heat originally supplied, W the work produced and Q2 the heat rejected, we have
Q1 = Q2 + W
The efficiency of the engine is
W/Q1
It is important to realize that Q2 is a necessary consequence of the second law of thermodynamics, which states that heat cannot be completely converted into work.
The sum of the effects of our technology is that of a heat engine which still depends largely on fossil fuels but which may be in the process of conversion to nuclear fuel, should this prove socially acceptable.
It is interesting that although we speak of a quantity of useful work W, and the corresponding thermal efficiency, neither of these concepts has any direct meaning as far as the environment is concerned. First of all it is clear that W depends on the way it is measured and on what we regard as useful work. The environment, on the other hand, sees only Q1 , all of which must be dissipated or stored. Thus in boring a hole in a steel block, we expend Q1, obtain W and reject Q2. However W is transformed to other energy forms such as the heat of friction, which must be dissipated. This heat may appear as thermal pollution in addition to that produced by Q2. We may then ask what effect on the environment does efficiency have? The answer is that the effect of an increased W/Q1 is to require a smaller Q1 to accomplish any given task that we regard as "useful." The smaller the Q1, the smaller the quantity of pollution and environmental impact for any given process. Thus the environmental impact of engine efficiency depends not on W/Q1, but on Q1. An obvious conclusion, which is not exactly new, is that the environment would benefit from the absence of technology.
In thermodynamics it is always necessary to define the system to which reference is made. In our examples we shall be concerned with systems of varying size, and all these systems shall be "open" in that some sort of communication will exist with the surroundings. The smallest type of system we shall consider is a restricted region of Earth's surface of the order of a kilometer or less in dimension and with a vertical dimension of the order of several kilometers, including the subsoil and overlying atmosphere. We shall consider particularly the energy flux through such regions before and after they are modified by technology. The largest system of interest is the entire surface region of the planet, including the atmosphere and oceans. This region, encompassing the entire biosphere, is the region of ultimate pollution dissipation and the region penultimate energy dissipation. The biosphere is practically closed with respect to pollutants themselves, but as these become increasingly degraded, their energy is radiated into space.
It is apparent that the technological engine is very inefficient as estimated from its components, no matter which way this efficiency is measured. Even the most advanced electric power generating plants have thermal efficiencies of less than 40 percent and most power plant efficiencies are nearer 30 percent (Barr,1970). Our dominant transportation systems are far less efficient than this. For example, it has been estimated that the private automobile has an efficiency of less than 10 percent, although only about one passenger is transported (Freeman, 1970). However, we have seen that pollutants, whether they be of a physical or chemical nature, are bearers of excess energy.
If this energy is dissipated by direct transfer of heat, we refer to it as thermal pollution. If it is dissipated by exothermic chemical reaction of unstable synthetic compounds, we call it chemical pollution, and if by energetic particles and fluids we call it physical pollution.
In addition to the heat dissipated in pollution there are also the effects of changes of entropy. Entropy is an index of the degree of disorder present in matter. The entropy always increases with an increase in the disorder or randomness of atomistic particles. Such an increase in disorder is inherent in spontaneous processes such as heat flow and chemical dissolution. Thus the net effect of heat dissipation from a heat engine is an increase of entropy for the total system of engine and the surrounding environment. The same is true when atoms, ions or other particles dissolve into the environment. For example, the dispersion of mercury or other toxic metals into natural waters, bottom sediments and organisms is difficult to prevent or counteract because of the large increase in entropy of mixing involved in the dispersion.
Pollutants emanate from natural environments which have been modified by technology and they carry off the excess energy of these environments. Modified environments therefore exist at higher levels of energy absorption and dissipation than the natural environments which prevail under the given geological and climatic conditions. If we confine ourselves to the case in which no local energy sinks or sources are involved, and in which a steady state prevails, the general picture may be outlined as in Figure 1. In this simplified and idealized scheme a given quantity of energy flows into the system and the same quantity flows out during the average yearly cycle. Figure 1-a shows the natural "ground state" in which there is a certain internal conversion of energy forms within a restricted environmental region of the Earth's surface which depends on details of the geology, climate and the specific ecology. For example, solar radiant energy is utilized both in photosynthesis and in heating the water in the run-off. The region of interest is regarded as extending upward to a "compensation level", which is defined as that level of the atmosphere above which a given environmental modification has a negligible effect on the total weather energy input and output. For example, replacing a forest by a grassy field would decrease the interaction between air and the surface through diminished roughness. However, this effect probably would extend only a few thousand feet above the site. On the other hand, dense smoke emissions from an industrial site could cut off considerable light to higher elevations. Strictly, such compensation surfaces should be included for subsoil and lateral exchange; however, these are probably minor effects in most cases.
Figure 1-b shows the technological energy input and the equivalent energy dissipated outward in a modified region. This figure refers to the simple case of an "isolated modified region."
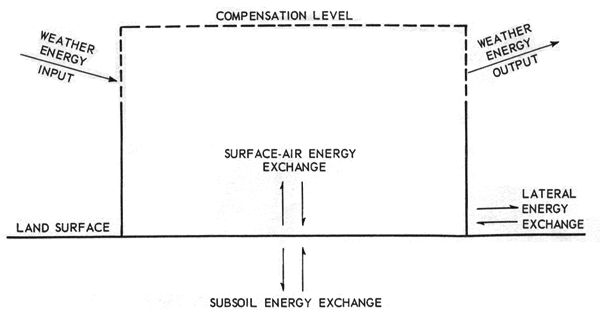
(a) Natural Region or "Ground State"
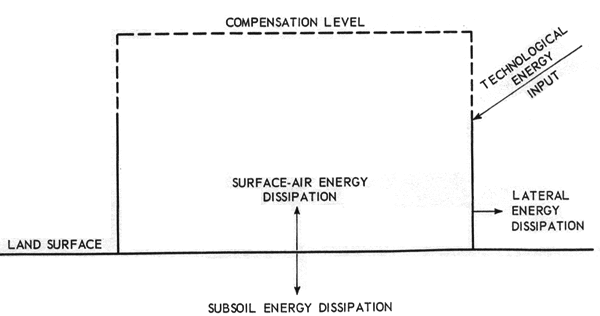
(b) Isolated Modified Region Component for Superposition on (a)
Figure 1. Modifications of Natural Region by Technology
Isolated regions are distinguished from non-isolated regions by the characteristic that the former emit pollutants but absorb virtually none from other regions. An example of an isolated region is a cleared field maintained as a meadow in a large forest tract. It is obvious that a region in a city does not qualify as an isolated region since it receives pollutants from nearby regions.
Figure 1-b should be regarded as being superimposed on Figure 1-a. Then if the compensation level (surface) choice is adequate, the total original energy component exchanged with the external environment is the same as before modification, except that the energy spectrum will in general be altered, and the additional technological energy received must be dissipated into the surroundings.
The modified regions of high energy dissipation are analogous to atomic and macroscopic chemical systems in that these regions spontaneously revert to the ground or low energy state of the indigenous ecosystem if the technological energy is withheld. We may illustrate by considering a typical land area of the northeastern United States. Under natural conditions such an area will be covered by a climax forest consisting largely of hardwoods. If this forest is cleared and planted to grass, a continual input of energy is required to maintain it as a meadow. The power absorbed by the meadow and dissipated in various ways is a definite quantity in excess of that dissipated by the climax forest, the atmosphere above and the underlying soil under natural conditions. The excess energy will be dissipated by such pollutants as silt, smoke, sound, pesticides, fertilizers, etc., which wash into the streams or which escape into the air.
It should be pointed out that at least three secular changes result in deviations from the steady state situation. These are
It might also occur to some readers that mass should serve as an alternative to energy as a characteristic of pollutants, since mass units are frequently employed in pollution monitoring. However, brief consideration will show that there is no simple relation between mass and energy content above the size range of subatomic systems. A good example is radioactive pollutants, in which only a very small mass may be involved. It therefore appears that mass is a parameter of less general applicability than energy in comparing one pollutant with another.
At this point it might also be asked why high energy content of pollutants should so frequently have deleterious effects on the organic world. It must first be understood that there is nothing wrong with energy per se, since all biological systems utilize energy in some form or other. However, we are here concerned with energy that is spontaneously dissipated into the surroundings and therefore corresponds to a large increase in the entropy or disorder of the environment. This type of energy leads to particles being in random locations at unforeseen times and on random breakage or formation of chemical bonds. Organisms, on the other hand, require energy of certain definite types in certain spectral ranges. Furthermore, they have adapted to absorb this energy from familiar sources such as sunlight. There is little in their genetic heritage that enables them to cope with the great variety of pollutant energy sources.
ENERGY CONCENTRATION IN MODIFIED REGIONS
Although the excess energy flux through modified regions in a steady state balances the technological energy input, there may also be conspicuous concentrations of energy from existing natural sources, and this energy may greatly enhance the flow of pollutants. This occurs, for example, when natural water run-off is accelerated by removal of vegetation. The energy of falling water is then concentrated in time and in particular localities, with resulting sheet wash and gully erosion. As is the case with excess technological energy, the energy concentrated in this way is frequently out of harmony with the existing ecology and with man's own goals. Indeed, the large drainage modification projects for hydropower and flood control, are also of this kind. It is well known that the long term disadvantage of such energy concentrating projects in many cases outweigh their short term benefits.
RECYCLING, POLLUTION CONTROL AND LIFE STYLE
The concept of the recycling of goods and the utilization of "pollution control devices" have found great favor among the general public and even among environmentalists. The attraction of these methods of pollution abatement stems largely from their apparent promise of being effective while allowing a continued high goods consumption rate or even permitting this rate to increase. However, these concepts, which may be characterized as "high energy concepts of pollution control", contain some serious contradictions from a thermodynamic standpoint. One such contradiction involves recycling. Perhaps the most prevalent view is that efficiency in recycling consists in recovering the largest possible proportion of goods used. However, recycling is an attempt to save resources and to avoid the disposal of pollutants, and as such is an ordering process akin to manufacturing itself.
Such processes also involve spontaneous thermodynamic processes of wear and energy dissipation and thus require an input of energy. Thus, although recycling may indeed reduce the total consumption of energy and other resources, this result is by no means assured. In any case, recycling involves the substitution of one form of pollution for the original form. For example, the recovery of glass bottles may substitute detergent and combustion pollution for glass pollution. All these factors must be given proper weight in determining the efficiency of a recycling process. An instance of the law of diminishing returns is the hypothetical 100 percent recovery of some recycled item. It is clear that such an attempt would involve the prevention of wear and so would require an infinite input of energy and would create an infinite amount of pollution. It is not difficult to think of practical instances in which this principle is operative-for example, in the recovery of bricks during building demolition.
Equally important illustrations are provided by pollution control devices such as are installed on automobiles or industrial plants. As in the case of recycling, such devices are intended to allow a given technological activity to continue undiminished. However, analysis of the type presented in the previous sections suggests that the hoped-for "clean" cars and industries may be virtually indistinguishable from their "dirty" predecessors in overall effect. In the case of the automobile the proposed pollution control devices are largely directed at the elimination of high energy gaseous emissions such as CO, hydrocarbons and nitrogen oxides. However, there is a very large and as yet undetermined amount of pollution which results from the manufacture, wear, servicing, and cleaning of automobiles. The considerable quantity of oil which drips from cars in parking lots and on highways finds its way into streams and storm drains and is a major stream pollutant. Also, the direct and indirect stream, land and air pollution which results from highways and from highway material manufacture is in the early stage of documentation (Mueller and Lahn, 1970). The proposed emission control programs are well known to be associated with an expanding automotive technology which will lead to an increase of most of the pollutants not covered by control devices. Part of the expanded technology will also be associated with the control devices and their monitoring systems. If this overall expansion to technology is not accompanied by a compensating increase in efficiency, there will be a net increase in pollution as a result of the installation of the control devices.
In the past the efficiency of a technological process, whatever the criteria of efficiency, has almost invariably been confined to the process itself. By this method it has been possible to build "sophisticated technologies" by utilization of a far greater base of unsophisticated and inefficient technology. However, in environmental problems we are concerned with the total planetary system. Under these circumstances we are literally faced with either increasing the efficiency of the technological engine from the inside or curtailing its operations.
Thus we have the choice before us of two different types of technological processes. The first type, the high energy process, has already been shown to contain contradictions. It has never been demonstrated, to the writer's knowledge, that such a process, as exemplified by recycled goods, or installation of pollution control devices, makes possible an overall abatement of pollution. The second type of process may be called an "economy process of pollution abatement." Such a process does involve a fundamental reduction of both resource consumption and pollution. However, it may also result in inconveniences, since it depends on diminished goods and services. A good example is the substitution of public mass transportation for the private automobile. Another example is the elimination of luxury goods, which are the objects of recycling processes and the substitution of goods of a more frugal nature. It seems unlikely that the high energy pollution abatement methods will accomplish their objectives, since they depend upon an undiminished flow of energy, and this energy must in some way be dissipated.
ECONOMICS AND THERMODYNAMICS
It has been pointed out (Boulding, 1968; Beers, 1970) that the dominant economic philosophy of our times, as distinguished from the science of ecology, has developed an open system approach that takes no account of the ultimate disposal of the resources utilized by technology. Ecology, on the other hand, stresses the need for balance and nearly closed cycles in resource utilization. It is concluded here that not only general economic policy but also the high energy concept of pollution abatement is incompatible with the ecologic view and ignores the best known thermodynamic laws. We must therefore search for economic policies which are in closer harmony with the cyclic system of nature and which seek to minimize the energy flux, which feeds technology.
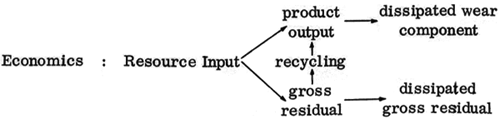
An essential parallelism between the resource flow scheme of economics:
and the energy flow scheme of thermodynamics:
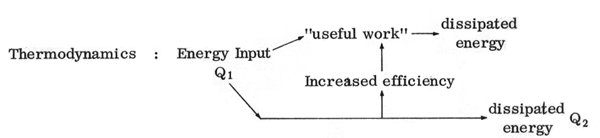
is apparent. The term "gross residual", taken from Cumberland et al (1970), is defined as all by-products of production which are waste materials. We observe that just as there is dissipated energy component Q2, which may be diminished by increased efficiency, so there is a gross residual of production, which may in part be reclaimed. Similarly to the case of useful work or energy the product output is eventually entirely dissipated into the environment as "wearout." Just as energy becomes increasingly unavailable toward the right in the diagram, so also the value of goods are diminished as they wear out.
To the present time technological processes have depended on "cost avoidance" (Olson and Cumberland, 1970), by which goods are underpriced to the extent that they do not include the cost of their disposal in the environment. Generally the rejected goods, including the pollutants, have been disposed of in the public domain. New economic policies should demand that marketed goods include these costs in their prices. In addition to paying for direct pollution abatement, such a policy would have the effect of increasing the value of goods so they would not be falsely rejected before they are worn out. It is obvious that this would reduce total resource consumption. It is also obvious that such policy clashes seriously with our dominant life style.
SUMMARY AND CONCLUSIONS
The technology of man may be regarded as a heat engine and as such is subject to thermodynamic principles which govern energy transformations. In this context pollution in its myriad forms is seen as the agent by which the total energy input is dissipated into the environment. Pollution source regions are classified as isolated and non-isolated regions of our planet that have been modified by technology. Such regions are distinguished by higher levels of energy flux than the natural regions from which they were derived. Technologically modified regions revert spontaneously to natural regions if the technological energy is withheld and provided no permanent modifications have occurred. In this scheme thermal efficiency has pertinence only inasmuch as it enables the total energy input to be reduced. In this scheme pollution is seen as the inevitable consequence of the technological energy flux to which the organic world is not adapted.
Pollution abatement programs are classified as either "high energy" or "economy" processes. Today high energy processes of abatement, which are characterized by an undiminished energy flux and expanding technology, are much in the vogue. Typical of these are the proposed extensive pollution control technologies, which rely on pollution control devices. By contrast, the economy processes, which depend on a diminished energy flux through diminished goods and services and which incur inconveniences, are unpopular.
The thermodynamic approach to environmental problems has its parallel in economics. This parallel is clearly revealed in economic policies that take into account the ultimate disposal of goods and services by including the cost of this disposal in the initial price of goods. It is likely that the recognized parallelism also reflects the underlying thermodynamic basis for such policies.
The thermodynamic analysis of technology exposes the need for comprehensive resource expenditure inventories, which encompass every aspect of a given project. Such an inventory must take into account the gross residual and wear components as well as the product output. In the case of pollution abatement projects precise energy expenditure inventories, which cover every aspect of technological expansion, must be undertaken to determine whether the control program will actually result in an overall decrease in pollution. It is also clear that the system of interest in such inventories is the entire planet. Since technological energy is dissipated throughout the biosphere, a search must be made for energy dissipation paths that lead to the least environmental stress. Unfortunately, technological expansion which is in response to narrow goals is not likely to yield energy forms which are harmlessly dissipated. Consequently, it seems advisable to seek economy forms of pollution abatement in order to minimize the energy flux.
The need for economy processes in both resource utilization and in pollution control is also brought out by recent resource inventories. For example, Hubbert (1969) has shown that the curves of production rate versus time for the major classes of fossil fuels have a characteristic bell-shape with distinct maxima which will occur in the near future. For example, the production rate peak of natural gas from the United States and adjacent continental shelves exclusive of Alaska is estimated to occur about 1980. This type of analysis is likely to have an even more pressing relevance in the future, especially since the production of nuclear energy has so many environmental uncertainties.
REFERENCES
Barr, W. A. (1970). Intergovernmental Coordination of power development and environmental protection act (S. 2752) Hearings before subcommittee on intergovernmental relations, Ninety-first Congress, Part I, 285-301.
Beers, R. F. (1970). Intergovernmental Coordination of power development and environmental protection act (S2752), Hearings before subcommittee on intergovernmental relations, Ninety-first Congress, Part I, 242-268.
Boulding, K. E. (1968). Beyond Economics, University of Michigan Press.
Commoner, Barry (1969). Pollution of the environment. Colloquium at Goddard Space Flight Center, Greenbelt, Maryland, 17 October, 1969.
Cumberland, J. H., Gerhardt, Paul, Gibson, C. S. Jr., Hibbs, J. R., Korbach, R. J. and Strain, B. N. (1970). EQUIPS-MABET, Preliminary demonstration phase of the environmental quality information and planning system (EQUIPS). The effort is oriented toward the development of a material balance externality trace (MABET) model. Preprint, University of Maryland.
Freeman, S. D. (1970 ). "Energy use and environmental protection- issues for the regulators". Speech to annual convention National Association of Regulatory Utility Commissioners, Los Vegas, Nevada. ( Director Energy Policy Staff, Office of Science and Technology, Executive Office of the President, Washington, D. C. ).
Hubbert, M. K. ( 1969 ). Energy resources. in Resources and Man, W. H. Freeman and Co., San Francisco Ca.
Kubaschewski, O. and Evans, E. LL. ( 1958 ). Metallurgical Thermochemistry, Pergamon Press, New York, N. Y.
Mueller, R. F. and Lahn, R. M. ( 1970). The Anacostia River, Ecological Imbalance of an Urban Stream Valley. NASA X-644-70-446.
Olson, C. E. and Cumberland, J. H. (1970 ). Intergovernmental Co-ordination of power development and environmental protection act ( S. 2752 ), Hearings before subcommittee on intergovernmental relations, Ninety-first Congress, Part I. 226-237.
Raymahashay, B. C. (1968 ). A geochemical study of rock alteration by hot springs in the Paint Pot Hill area, Yellowstone Park, Geochim. et Cosmochim. Acta, 32, 499-522.
The Cost of Clean Water and its Economic Impact ( 1969 ). The Report, U. S. Department of Interior, Federal Water Pollution Control Administration.
Urone, P. and Schroeder, N. H. ( 1969 ). SO2 in the atmosphere: a wealth of monitoring data, but few reaction rate studies. Environmental Science and Technology 3, 436-445.